Empirical formula chemistry calculator2/20/2023  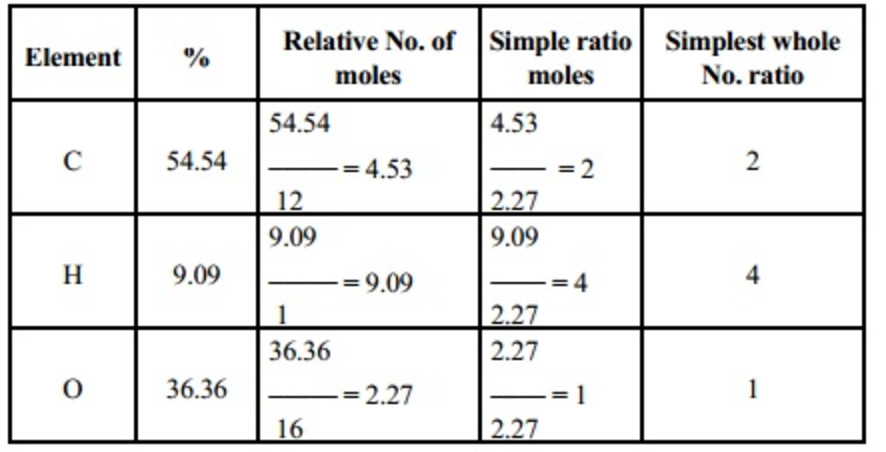
Step 6 Determine how many times greater the molecular mass is compared to the mass of the empirical formula. The formulas to keep in mind are: multiple of empirical formula molecular mass /. (4 carbon x 12.0) + (5 hydrogen x1.0) + (1 oxygen x 16.0) + (2 nitrogen x 14.0) =97.0g/mol Multiply each subscript in the empirical formula by the answer in step 2. Step 5 After you determine the empirical formula, determine its mass. The values are all really close to whole numbers. to get an integer is they are not all whole numbers Step 3 determine the mole ratio by dividing each elements number of moles by the smallest Step 1 Assume a mass of 100g so % becomes grams (Hint-Save the molar mass 194.19g/mol until the end) What is the molecular formula of caffeine? Step 7 Multiply the atoms in the empirical formula by this numberĮxamples- Caffeine has an elemental analysis of 49.48% carbon, 5.190% hydrogen, 16.47% oxygen, and 28.85% nitrogen. Index of all my online chemical calculation quizzes. Step 6 Divide the given molecular mass by your E.F. Using moles to calculate empirical formula and deduce molecular formula study examples carefully. Step 5 Determine the mass of your empirical formula The question should have included a molecular mass. Molecular Formula calculator uses Molecular Formula Molar Mass/Mass of Empirical Formulas to calculate the Molecular Formula, Molecular Formula is the. Step 4 Double, triple … to get an integer if they are not all whole numbers 
Step 3 Determine the mole ratio by dividing each elements number of moles by the smallest value from step 2. Step 2 Determine the moles of each element. If the data does not fit to a simple formula, the program will attempt to generate possible empirical formulae and will indicate how well these fit the percentage composition using the variance. Assume the mass to be 100g, so the % becomes grams. Enter the atomic symbols and percentage masses for each of the elements present and press 'calculate' to work out the empirical formula. Step 1 If you have masses go onto step 2. Level 2 Empirical Formula Calculation Steps What is the empirical formula of the following compounds? (so reduce the formula if you can) molecular formula 
Level 1 Simple Empirical formula questions Empirical and Molecular Formula CalculationsĮmpirical formula is the smallest whole number ratio of moles of each element in a compound.ĬaCl 2 -> there is 1 mole of calcium for every 2 moles of chlorine 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
